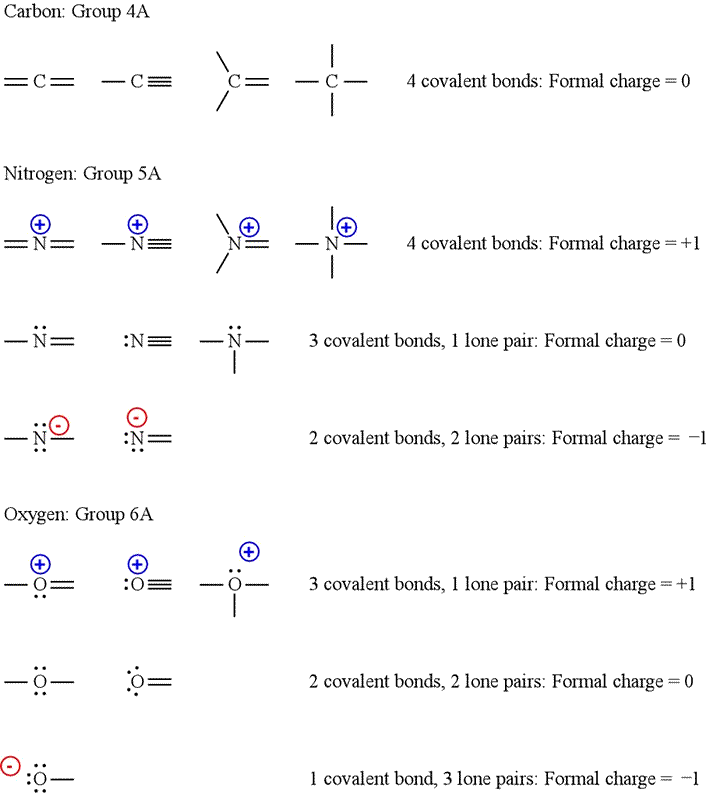
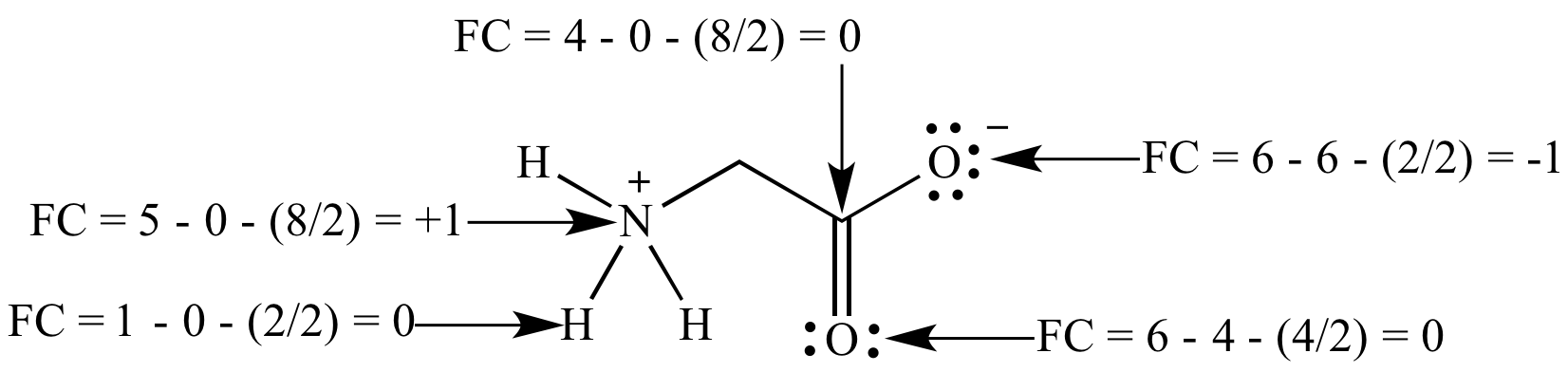
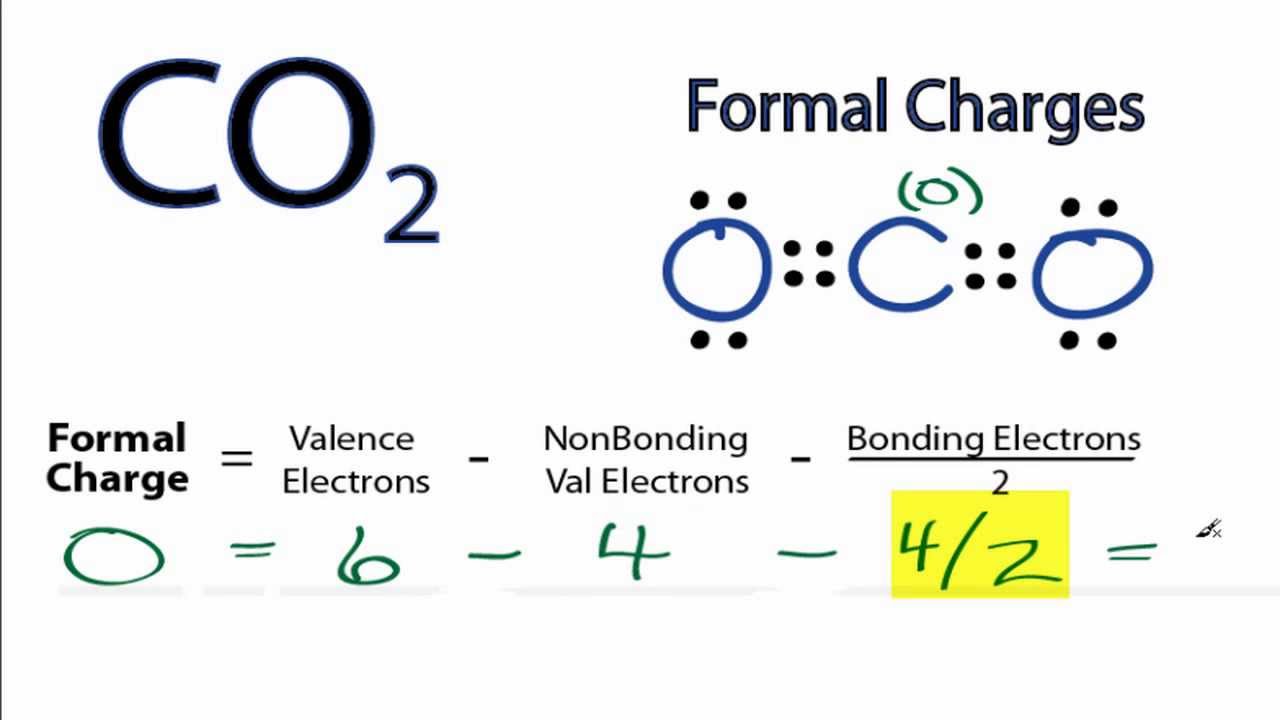
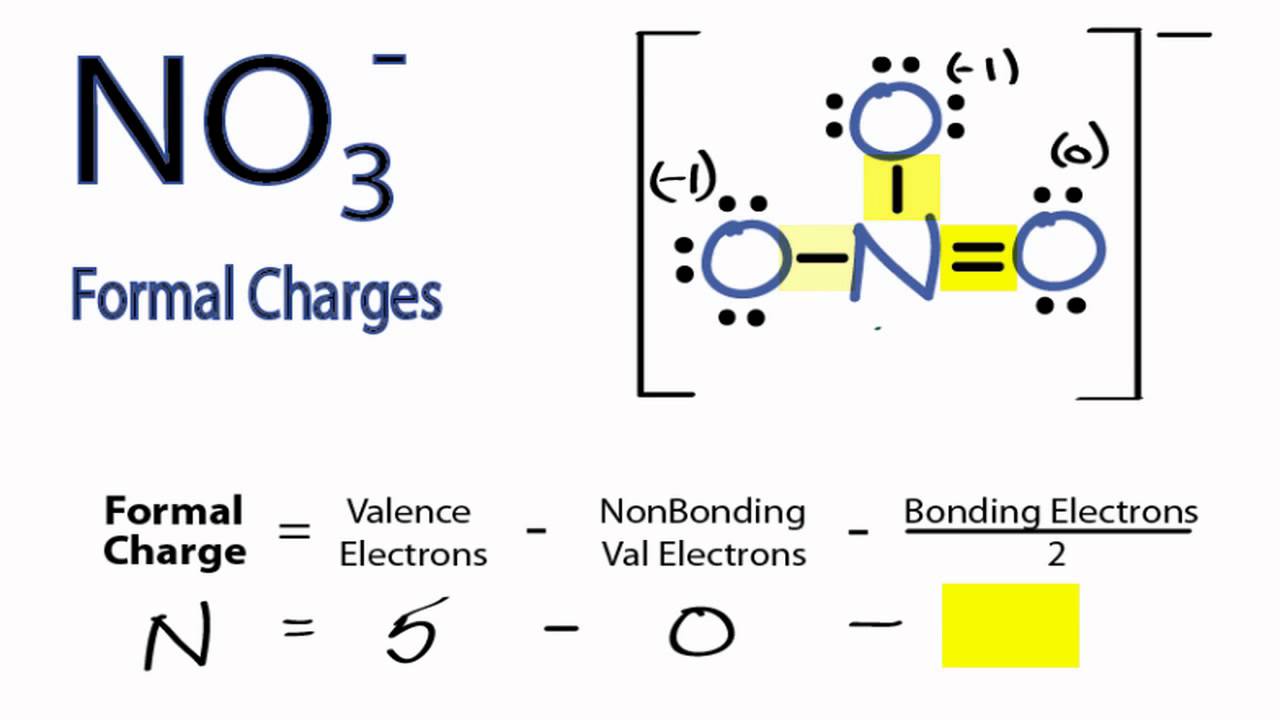
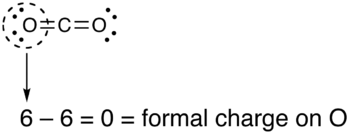
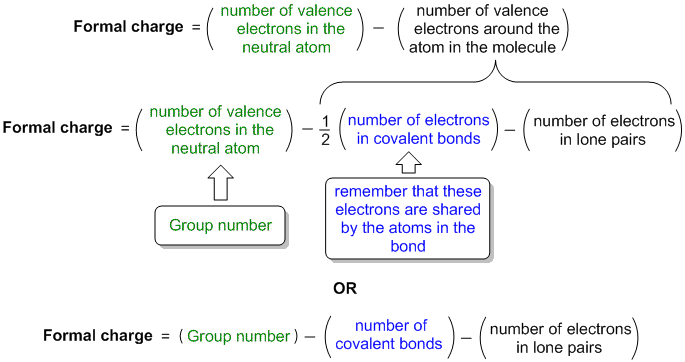
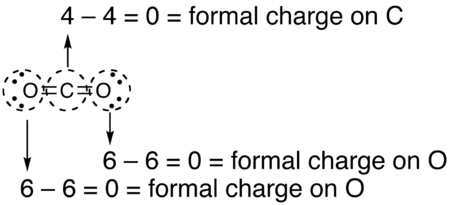
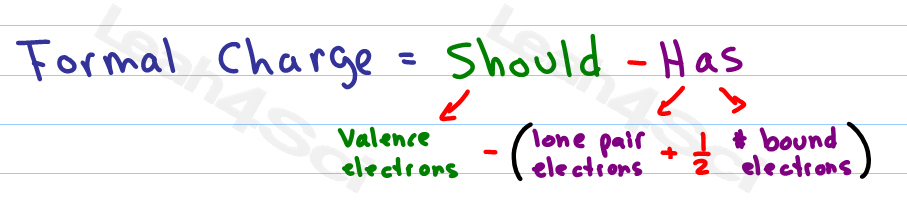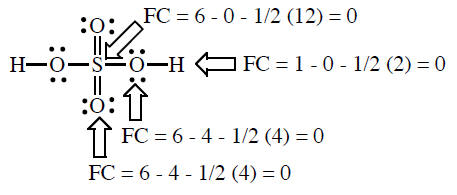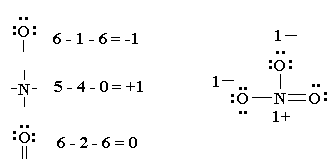formal charge
We offer you for free download top of formal charge pictures. On our site you can get for free 20 of high-quality images.
For your convenience, there is a search service on the main page of the site that would help you find images similar to formal charge with nescessary type and size.